As pollutants cased from base smelting rise into the atmosphere , such as sulfides produced from copper smelting, they react with H2O molecules in the atmosphere to create acid rain ("Effects of Acid Rain")
H2O(l) + SO2(g) ==> H2SO3(g)
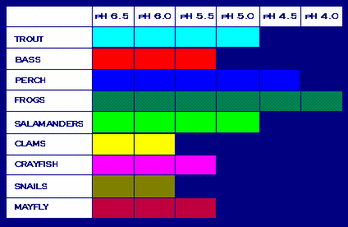
*charts shows certain species toxicity tolerance
H2O(l) + SO2(g) ==> H2SO3(g)
- This acid rain than falls to earth down to earth ("Effects of Acid Rain")
- This rain falls onto forests, agriculture fields, roads, building and other areas before flowing into streams, rivers and lakes ("Effects of Acid Rain")
- It may also fall directly on top of aquatic habitats ("Effects of Acid Rain")
- All of this will increase the acidity of the lakes, rivers and other bodies of waters which may dramatically affect the the species of aquatic organisms that live there ("Effects of Acid Rain")
- Aluminium levels also increase in bodies of waters where the buffering capacity of the surrounding soil is low, this releases aluminium from the soil ("Effects of Acid Rain")
- Increased acidity and aluminium levels may result in direct decrease of the fish population however some aquatic species are more tolerant to increased acidity than others this can mean several things ("Effects of Acid Rain")
- That some species will decrease because although some species are resistant to toxicity at full maturity they may be highly sensitive at a young age for example- certain species of fish can stand increased toxicity levels but fish eggs can not hatched at a pH level of 5.("Effects of Acid Rain")
- Although certain species may not be directly affected my increased acidity, species that they depend on for food may be. For example frogs may not be affected because they have a high tolerance for acidity but if they eat insects like mayfly part of their food supply will disappear because mayfly can not survive increased toxicity. ("Effects of Acid Rain")
*charts shows certain species toxicity tolerance

